Structure: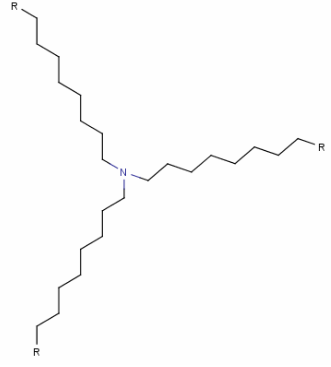
| 外観 | Colorless clear liquid |
| 式 | C27H57N |
| CAS番号. | 68814-95-9 |
| EC番号. | 214-242-1 |
| いいえ. | 3082 |
| HSコード | 2921199090 |
| Tertiary Amine % | ≥98 |
| Tertiary amine nitrogen content | ≥3.3 |
| Acid value(mg KOH/g) | 128-142 |
| Typical carbon chain % | C8 :35%-42% C10:52%-63% |
| 密度(20℃)g/ml | 0.81 |
| 引火点℃: | ≥206 |
| 同義語 | N235 7301 |
Trialkyl amines are typically colorless liquids or solids at room temperature, depending on the
length and nature of the alkyl groups.
Trialkyl amines often have a strong, fishy odor, especially in the case of lower alkyl chain lengths.
Trialkyl amines are generally soluble in organic solvents such as ethanol, methanol, and chloroform,
but their solubility in water varies depending on the alkyl groups. Smaller alkyl groups increase water
solubility, while larger alkyl groups decrease it.
Trialkyl amines are weak bases and can undergo protonation reactions. The basicity of a trialkyl amine
depends on the alkyl groups and their electron-donating or -withdrawing nature. Generally, tertiary
amines (trialkyl amines with three alkyl groups) are more basic than secondary or primary amines.
Trialkyl amines can participate in a variety of chemical reactions. 例えば, they can undergo alkylation
reactions where the alkyl groups are modified or substituted. They can also react with acids to form salts,
and they can be oxidized to form amine oxides.
The boiling point of a trialkyl amine depends on the size and nature of the alkyl groups. Generally,
trialkyl amines with longer alkyl chains have higher boiling points due to increased intermolecular forces.
Trialkyl amines are often used as catalysts in chemical reactions. They can facilitate reactions such as
esterification, transesterification, and Michael additions. Tertiary amines, in particular, are known for
their catalytic activity in various organic transformations.
Trialkyl amines are used in the synthesis of pharmaceutical compounds. They can be used as building
blocks or intermediates in the production of drugs, such as antihistamines, antidepressants, antivirals,
and local anesthetics.
Trialkyl amines can be employed as corrosion inhibitors to protect metals from degradation caused by
exposure to corrosive environments. They form a protective layer on the metal surface, preventing or
reducing corrosion.
Trialkyl amines can be used as surfactants, which are compounds that lower the surface tension between
two substances. They are utilized in various applications, including detergents, emulsifiers, fabric softeners,
and foaming agents.
Trialkyl amines can be incorporated into polymers as additives to enhance their properties. They can
improve the flexibility, adhesion, and thermal stability of polymers, making them suitable for applications
such as coatings, adhesives, and sealants.
Trialkyl amines are utilized in gas treatment processes, such as gas sweetening and carbon dioxide (CO2)
capture. They can absorb acidic gases, like hydrogen sulfide (H2S) and CO2, from natural gas or industrial
emissions, helping to purify the gas stream.
Trialkyl amines are versatile reagents in organic synthesis. They can participate in reactions like reductive
amination, acylation, and nucleophilic substitution, enabling the formation of various chemical compounds.
Trialkyl amines can act as initiators or co-catalysts in polymerization reactions. They can facilitate the
formation of polymers through processes such as radical polymerization or coordination polymerization.
The primary step is to choose the appropriate primary or secondary amine, which will be alkylated to form
the desired trialkyl amine. The alkylating agent, such as an alkyl halide (e.g., alkyl bromide, alkyl chloride)
or an olefin (e.g., alkene), is also selected based on the desired alkyl groups to be incorporated.
The alkylation reaction involves the reaction of the primary or secondary amine with the alkylating agent.
The reaction can be carried out under different conditions, such as in the presence of a solvent or a catalyst.
The reaction conditions may vary depending on the specific reactants and desired product.
After the alkylation reaction, the crude reaction mixture is typically purified to separate the desired
trialkyl amine from any unreacted starting materials or side products. Purification techniques such as
蒸留, 抽出, or chromatography may be employed to obtain a pure product.
The synthesized trialkyl amine is characterized using various analytical techniques, such as spectroscopy
(e.g., NMR, IR) and chromatography (e.g., GC, HPLC) to confirm its identity and purity.
• Incoming inspection: 主な原材料,内容が検査される, appearance and other
main properties.
• Feeding inspection: 原料供給の原則は先入れ先出しです, and the appearance of the
main raw materials is randomly inspected according to whether there is a big change in the storage
conditions before feeding.
• Batch sampling in the production process: 生産工程中, the main indexes of each batch
of products: 含有量と酸価は異なる期間で3回検査されます.
• Storage Inspection: 各バッチ (4トン) 保管前に検査されます.
• Outbound inspection: 顧客の要求量に応じて, the products will be sampled
and inspected.
• Pre-shipment inspection: 顧客の要求に従って, 第三者による検査が可能
出荷前の製品に.
• Physical Appearance: 外観検査
• Purity Analysis:ガスクロマトグラフィー (GC)
• Acid Value:電位差滴定
• Water Content:水分計
• Refractive Index:屈折計
• Density:密度計
製品の各バッチには適合証明書が添付されている必要があります, 含む: の名前
メーカー, 製品の名前, 製造バッチ番号, バレルあたりの正味重量, the
quality level and the implementation of the standard number.weight, quality grade and implementation
standard number.
清潔で乾燥したプラスチックドラムに梱包, 正味重量 ドラムあたり 200±0.3kg または 1000±0.5kg, 圧縮されて
各バッチ後に密封される.
この商品はプラスチックドラムに梱包されています, 輸送時や積み下ろし時, それは慎重に行うべきです
そしてそっと下ろします, そして衝撃を防ぐ.
保管場所は涼しい場所である必要があります, 乾燥していて換気されている. 耐火性と防雨性を備えています.

というコンセプトを追求している会社です。 “継続的な革新, 一流の追求”, 国内外の顧客に高品質の製品と満足のいくサービスを提供したいと考えています.