Structure: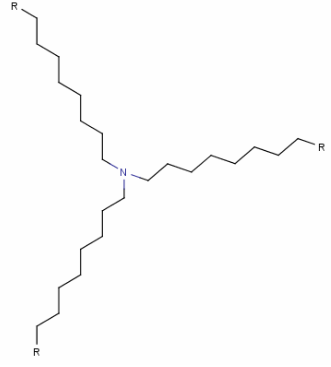
| 모습 | Colorless clear liquid |
| 공식 | C27H57N |
| CAS 번호. | 68814-95-9 |
| EC 아니요. | 214-242-1 |
| 아니요. | 3082 |
| HS 코드 | 2921199090 |
| Tertiary Amine % | ≥98 |
| Tertiary amine nitrogen content | ≥3.3 |
| Acid value(mg KOH/g) | 128-142 |
| Typical carbon chain % | C8 :35%-42% C10:52%-63% |
| 밀도(20℃)g/ml | 0.81 |
| 인화점℃: | ≥206 |
| 동의어 | N235 7301 |
Trialkyl amines are typically colorless liquids or solids at room temperature, depending on the
length and nature of the alkyl groups.
Trialkyl amines often have a strong, fishy odor, especially in the case of lower alkyl chain lengths.
Trialkyl amines are generally soluble in organic solvents such as ethanol, methanol, and chloroform,
but their solubility in water varies depending on the alkyl groups. Smaller alkyl groups increase water
solubility, while larger alkyl groups decrease it.
Trialkyl amines are weak bases and can undergo protonation reactions. The basicity of a trialkyl amine
depends on the alkyl groups and their electron-donating or -withdrawing nature. 일반적으로, tertiary
amines (trialkyl amines with three alkyl groups) are more basic than secondary or primary amines.
Trialkyl amines can participate in a variety of chemical reactions. 예를 들어, they can undergo alkylation
reactions where the alkyl groups are modified or substituted. They can also react with acids to form salts,
and they can be oxidized to form amine oxides.
The boiling point of a trialkyl amine depends on the size and nature of the alkyl groups. 일반적으로,
trialkyl amines with longer alkyl chains have higher boiling points due to increased intermolecular forces.
Trialkyl amines are often used as catalysts in chemical reactions. They can facilitate reactions such as
esterification, transesterification, and Michael additions. Tertiary amines, in particular, are known for
their catalytic activity in various organic transformations.
Trialkyl amines are used in the synthesis of pharmaceutical compounds. They can be used as building
blocks or intermediates in the production of drugs, such as antihistamines, antidepressants, antivirals,
and local anesthetics.
Trialkyl amines can be employed as corrosion inhibitors to protect metals from degradation caused by
exposure to corrosive environments. They form a protective layer on the metal surface, preventing or
reducing corrosion.
Trialkyl amines can be used as surfactants, which are compounds that lower the surface tension between
two substances. They are utilized in various applications, including detergents, emulsifiers, fabric softeners,
and foaming agents.
Trialkyl amines can be incorporated into polymers as additives to enhance their properties. They can
improve the flexibility, adhesion, and thermal stability of polymers, making them suitable for applications
such as coatings, 접착제, and sealants.
Trialkyl amines are utilized in gas treatment processes, such as gas sweetening and carbon dioxide (CO2)
capture. They can absorb acidic gases, like hydrogen sulfide (H2S) and CO2, from natural gas or industrial
emissions, helping to purify the gas stream.
Trialkyl amines are versatile reagents in organic synthesis. They can participate in reactions like reductive
amination, acylation, and nucleophilic substitution, enabling the formation of various chemical compounds.
Trialkyl amines can act as initiators or co-catalysts in polymerization reactions. They can facilitate the
formation of polymers through processes such as radical polymerization or coordination polymerization.
The primary step is to choose the appropriate primary or secondary amine, which will be alkylated to form
the desired trialkyl amine. The alkylating agent, such as an alkyl halide (e.g., alkyl bromide, alkyl chloride)
or an olefin (e.g., alkene), is also selected based on the desired alkyl groups to be incorporated.
The alkylation reaction involves the reaction of the primary or secondary amine with the alkylating agent.
The reaction can be carried out under different conditions, such as in the presence of a solvent or a catalyst.
The reaction conditions may vary depending on the specific reactants and desired product.
After the alkylation reaction, the crude reaction mixture is typically purified to separate the desired
trialkyl amine from any unreacted starting materials or side products. Purification techniques such as
증류, 추출, or chromatography may be employed to obtain a pure product.
The synthesized trialkyl amine is characterized using various analytical techniques, such as spectroscopy
(e.g., NMR, IR) and chromatography (e.g., GC, HPLC) to confirm its identity and purity.
• Incoming inspection: 주요 원료,내용이 검사됩니다, appearance and other
main properties.
• Feeding inspection: 원료 공급의 원리는 선입 선출입니다., and the appearance of the
main raw materials is randomly inspected according to whether there is a big change in the storage
conditions before feeding.
• Batch sampling in the production process: 생산 과정에서, the main indexes of each batch
of products: 함량과 산가는 서로 다른 기간에 세 번 검사됩니다..
• Storage Inspection: 각 배치 (4톤) 보관 전 검사를 거친다.
• Outbound inspection: 고객이 요구하는 수량에 따라, 제품은 샘플링됩니다
그리고 검사를 받았습니다.
• Pre-shipment inspection: 고객의 요구 사항에 따라, 제3자 검사를 수행할 수 있습니다.
배송 전에 제품에 대해.
• Physical Appearance: 육안 검사
• Purity Analysis:가스 크로마토그래피 (GC)
• Acid Value:전위차 적정
• Water Content:수분 분석기
• Refractive Index:굴절계
• Density:밀도계
각 제품 배치에는 적합성 인증서가 첨부되어야 합니다., 포함: 의 이름
제조업체, 제품 이름, 생산 배치 번호, 배럴당 순중량, 그만큼
quality level and the implementation of the standard number.weight, quality grade and implementation
standard number.
깨끗하고 건조한 플라스틱 드럼에 포장, 드럼 당 순중량 200±0.3kg 또는 1000±0.5kg, 압축하고
각 배치 후 밀봉.
이 제품은 플라스틱 드럼에 포장되어 있습니다., 운송 및 적재/하역 중, 조심스럽게 해야 해
그리고 천천히 내려놓으세요, 그리고 충격으로부터 방지.
보관장소는 서늘해야 한다, 건조하고 통풍이 잘되는. 방화 및 방수 처리.

회사는 다음의 개념을 추구합니다. “지속적인 혁신, 일류 추구”, 국내외 고객에게 고품질의 제품과 만족스러운 서비스를 제공하고자 합니다..